Sodium Ion Battery VS Lithium Ion—Safety
Release time:
2025-08-29
Share:
Sodium ion battery vs lithium ion is a highly talked-about topic in the battery market. Lithium-ion battery has dominated the new energy sector thanks to its high energy density. However, battery thermal runaway (TR) accidents raised many concerns about its safety. Does a sodium-ion battery, have the potential to replace lithium-ion batteries and be safer?

1. Are sodium batteries safer than lithium-ion? The answer is Yes.
According to the 2025 study Research on the Thermal Runaway Behavior and Flammability Limits of Sodium-Ion and Lithium-Ion Batteries (https://www.mdpi.com/2313-0105/11/1/24),
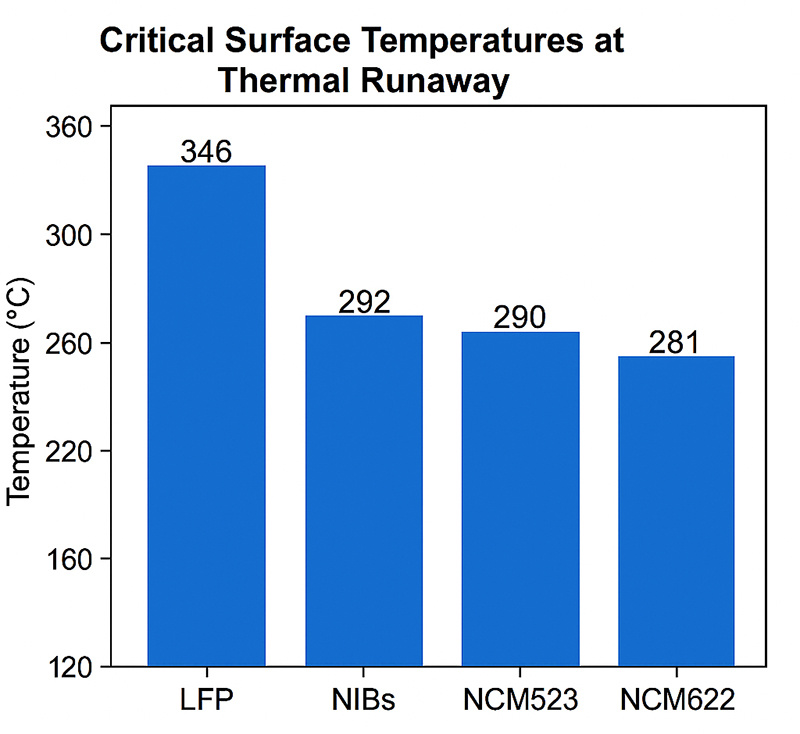
The critical surface temperatures on the heated side (Tf-TR) were ranked: LFP (346 °C) > NIBs (292 °C) > NCM523 (290 °C) > NCM622 (281 °C). That means sodium-ion batteries can tolerate higher temperatures before thermal runaway begins compared to common lithium-ion chemistries.
Sodium-ion batteries demonstrate higher thermal stability and a lower risk of catastrophic failure compared to lithium-ion batteries under laboratory abuse tests.
2. Why is sodium-Ion safer than lithium-ion batteries
There are three main reasons: higher thermal runaway temperature, slower thermal runaway processes, and fewer harmful gases.
1. Higher thermal runaway temperature
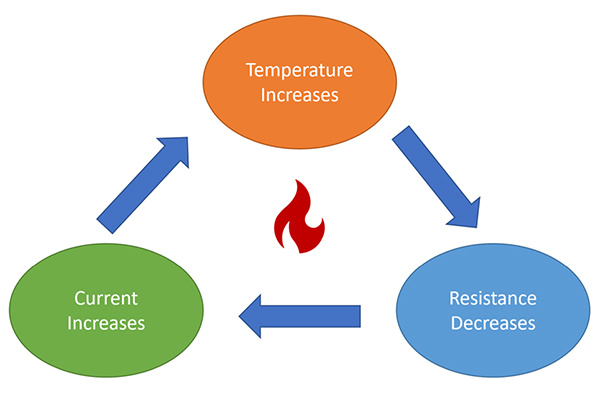
The thermal runaway temperature of sodium batteries is generally higher than that of lithium batteries. This means that in high-temperature environments, lithium batteries are more prone to thermal runaway, while sodium batteries are less likely to rapidly enter a dangerous state.
2. Slower thermal runaway process
The temperature rise rate during the thermal runaway process of sodium-ion batteries is slower than that of lithium batteries, and fewer flammable gases are produced after high-temperature decomposition. Although lithium-ion batteries have high energy density, they face thermal runaway risks caused by overcharging, overheating, short circuits, and physical damage, which may lead to uncontrollable fires or explosions.
3. Lower gas production and fewer harmful gases
In the event of thermal runaway, sodium-ion batteries release significantly fewer harmful gases than lithium batteries, and corrosive gases such as hydrogen fluoride are also less likely to be produced. This means that in an accident, sodium batteries pose a lower risk of harm to those in the vicinity.
3. Key Safety Aspects - Sodium Ion Battery Vs Lithium Ion Battery
|
Safety Aspect |
Lithium-Ion Battery |
Sodium-Ion Battery |
|
Thermal runaway temperature |
Lower. More prone to thermal runaway in high-temperature environments. |
Higher. NIBs (292 °C), with some cathode materials having even higher decomposition onset temperatures. |
|
Thermal runaway speed |
Faster temperature rise rates can lead to uncontrollable fires or explosions. |
Slower temperature rise rate; milder failure modes observed such as smoking rather than combustion in abuse tests. |
|
Gas production during thermal runaway |
Produces more harmful gases, including corrosive gases like hydrogen fluoride (HF). |
Releases significantly fewer harmful and corrosive gases. |
|
Risk of dendrite formation |
Lithium dendrites can pierce the separator, increasing internal short circuit risk. |
Sodium ions in the anode have significantly lower tendencies to form dangerous dendrites. |
|
Abuse test performance |
High failure intensity in abuse tests (needle penetration, overcharge, compression, thermal box). |
Higher thermal runaway triggers temperatures, slower propagation, and gentler failure outcomes under the same conditions. |
|
Material stability |
Requires complex internal/external safety mechanisms to compensate for lower material stability. Research is ongoing to improve thermal stability and reduce flammability. |
Material systems inherently have higher thermal stability. Industry is developing combinations with even better safety characteristics. |
|
Cost vs. safety balance |
High energy density but with more complex protection systems, increasing cost. Suitable for smartphones, laptops, etc. |
Lower cost and higher safety, making it suitable for energy storage scenarios where space and weight are not critical requirements. |
|
Real-world accident frequency |
Less than 1 accident per million high-quality cells, but the total battery quantity in use raises failure probability. |
Abuse tests and material characteristics indicate lower overall risk at scale. |
4. Lithium-ion battery safety
The main causes of thermal runaway in lithium batteries include overcharging, overheating, short circuits, and punctures. When a battery ruptures, is subjected to excessive external force, or experiences a malfunction in its control system, the stored energy can trigger thermal runaway, resulting in uncontrolled fires or explosions.
During the production of lithium-ion batteries, both external and internal protection mechanisms must be considered. External protection mechanisms are primarily achieved through components such as temperature sensors and pressure valves, but these increase the weight of the battery system and reduce its energy density. Internal protection mechanisms rely on the optimized design of the internal material components of the battery cell, focusing on the thermal stability, mechanical properties, and chemical properties of the materials to design high-safety battery materials, thereby fundamentally addressing the issue of poor safety in high-energy density lithium-ion batteries.
The lithium-ion battery industry has conducted extensive research aimed at improving the thermal stability of battery materials, reducing the flammability of battery compositions, and enhancing thermal management of battery modules. Thanks to these efforts, currently, fewer than one accident occurs per million high-quality lithium-ion batteries produced. However, whether used as a power source or for energy storage, hundreds or even thousands of lithium-ion batteries are required. As the number of batteries in use increases, so does the failure rate, which remains a major unresolved issue in lithium-ion batteries.

In October 2024, lithium-ion battery recycling plants suffered a massive explosion.
5. Sodium battery safety
Sodium-ion batteries offer significant potential in large-scale energy storage markets due to their abundant resources and low cost. However, beyond material costs, the more critical factor is that the inherent higher thermal stability of their material systems offers a new potential solution to the long-standing safety issues plaguing lithium batteries.
Although sodium batteries face the same thermal runaway triggers as lithium batteries (overcharging, overheating, short circuits, punctures), their material properties confer greater “resilience” when subjected to such abuses. For example, sodium battery cathode materials typically have a higher thermal decomposition onset temperature, and sodium ions at the anode exhibit significantly lower tendencies to form dangerous dendrites and puncture the separator compared to lithium dendrites, thereby reducing the risk of severe thermal runaway caused by internal short circuits from the source.
Industry research is continuously enhancing the safety characteristics of sodium batteries by developing material combinations with higher thermal stability. Results from numerous standard abuse tests (such as needle penetration, overcharging, compression, and thermal box tests) consistently show that, under identical conditions, well-designed sodium batteries exhibit higher thermal runaway trigger temperatures, slower temperature rise rates, and milder failure modes (such as only smoking rather than violent combustion) compared to lithium batteries. This strongly validates the inherent safety advantages of sodium batteries at the cell level.
 In large-scale applications (energy storage, power), numerous batteries are integrated. Lithium batteries face significant challenges in system safety design due to their high risk of thermal runaway and easy propagation. In contrast, sodium batteries have higher thermal stability and lower dendrite risks at the cell level, meaning each cell is less prone to thermal runaway. Even if a cell fails, its milder reaction and higher thermal propagation threshold make system-level thermal runaway propagation more difficult and slower, providing safety systems (such as insulation, fire suppression, and isolation) with longer response times and larger control windows. The inherent safety advantages of sodium battery internal material systems, combined with continuously updated external protection mechanisms (such as BMS), offer the battery industry a safe solution.
In large-scale applications (energy storage, power), numerous batteries are integrated. Lithium batteries face significant challenges in system safety design due to their high risk of thermal runaway and easy propagation. In contrast, sodium batteries have higher thermal stability and lower dendrite risks at the cell level, meaning each cell is less prone to thermal runaway. Even if a cell fails, its milder reaction and higher thermal propagation threshold make system-level thermal runaway propagation more difficult and slower, providing safety systems (such as insulation, fire suppression, and isolation) with longer response times and larger control windows. The inherent safety advantages of sodium battery internal material systems, combined with continuously updated external protection mechanisms (such as BMS), offer the battery industry a safe solution.
Sodium-ion batteries have demonstrated enormous potential in the large-scale energy storage market thanks to their abundant resources and low cost. But more importantly than material costs, the higher thermal stability of their material system offers a new possible solution to the long-standing safety issues of lithium batteries. This is also why the sodium-ion battery market has been booming in recent years.

FAQ:
Q: Why are sodium batteries less likely to catch fire?
A: 1. Materials are more heat-resistant (with 100°C+ higher decomposition temperature). 2. No dangerous dendrites form to pierce separators. 3. More stable, less flammable electrolytes
Q: Why are sodium batteries more suitable for large-scale energy storage systems?
A: Sodium-ion batteries are low-cost, offer enhanced safety and are less prone to fire under high temperatures or abnormal conditions. Although their energy density is slightly lower, volume and weight are not major constraints for stationary energy storage applications.
Q: Why do we see so many thermal runaway accidents in lithium batteries?
A: It always comes down to things like overcharging, overheating, or physical damage. These problems can trigger a chain reaction inside the battery and heat buildup finally leads to fire and explosion.
Q: Why are sodium batteries becoming more popular?
A: Two words: cheap and safe. And that's why sodium batteries are capturing the attention of the energy storage industry.
Previous Page:
FAQ
Latest Information